Stem cell therapy for MS
It is important for people with MS to have the best available information on stem cell therapy to make decisions related to this complex issue.
Last updated: 6th November 2025
What’s on this page?
There is continued need for more effective treatments for MS. Stem cell therapies are being explored as a potential way to address this unmet need. The unique properties of stem cells make them promising candidates for treatments that can slow MS disease activity.
There are different types of stem cells, and encouraging research is underway to determine how stem cells work, whether the use of stem cells to treat people with MS is safe and effective, and which forms of MS may benefit from different stem cell procedures.
It is important for people with MS to have the best available information to make decisions related to this complex issue.
What are stem cells?
Most cells in the body carry out very specific roles and are called specialised cells. Stem cells are different because they’re not specialised, which means they can develop into different types of cells, like nerve cells or immune cells. They are important as we grow and develop when young, and also to repair damage and replace cells as we age.
The types of stem cells being investigated as treatment options for MS are found in lots of different tissues, from bones to brains.
Making decisions about stem cell therapy
The term ‘stem cell therapy’ refers to any procedure that involves stem cells or their products. There are many different kinds of stem cells, and many types of stem cell therapies.
Most of these therapies are being studied in clinical trials. The only stem cell therapy that currently should be accepted as a treatment for MS outside of clinical trials is autologous Hematopoietic Stem Cell Transplantation (aHSCT), for which there is enough evidence about potential benefits, risks and side effects.
In addition to the stem cell therapies being developed specifically for MS, there are many other therapies involving stem cells that have never been trialled in MS. Experts have found that these unproven treatments are being marketed to people with MS, despite a complete lack of clinical evidence.
It is important for people to have the best available information to make decisions related to their MS treatment. Each person’s situation is unique and decisions about any MS treatments – taking into consideration the potential benefits, risks and side effects for an individual’s particular circumstances – should be made in careful consultation with each individual’s neurologist.
For any type of treatment, including aHSCT, it is important to make sure the clinics or centres offering the treatment are properly accredited, have substantial expertise in delivering the procedures, and have expertise in managing MS.
What types of therapy for MS use stem cells?
Autologous Hematopoietic Stem Cell Transplantation (aHSCT)
What is aHSCT?
Hematopoietic stem cells are a type of adult stem cell made in the bone marrow, which have the ability to produce the different cells found in the blood, including immune cells. In aHSCT, stem cells from a person’s own bone marrow are collected and stored, while a significant portion of the rest of an individual’s immune cells are reduced by chemotherapy. Following chemotherapy, the stored stem cells are reintroduced into the body and over time they produce new cells that repopulate the body with all types of blood cells including immune cells.
This treatment attempts to ‘reformat’ the person’s immune system, and stop the inflammation that contributes to active, relapsing MS or active, progressive MS. However, these stem cells are not able to regenerate damaged nerves or other parts of the brain and spinal cord.
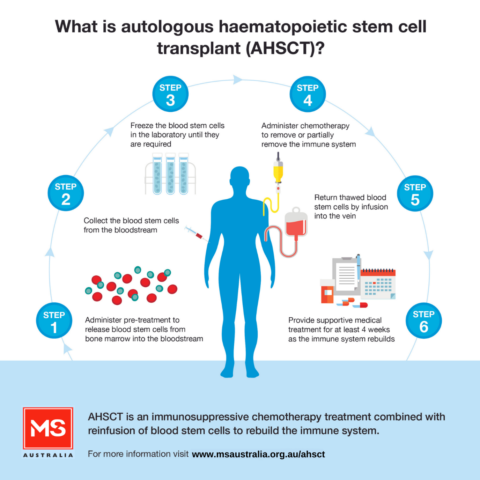
This diagram, showing the aHSCT process, comes from MS Australia.
MS is a very varied disease and therefore this treatment is not suitable for all people nor all types of MS. It is important to note that aHSCT is an intensive treatment with significant risks, including life-threatening for some people, and the chemotherapy itself has several known side effects.
Whilst aHSCT is considered to be a treatment for highly active MS in a few countries, the conditions under which it is made available may differ between, or even within, countries. In some cases, even if aHSCT is approved for use, it may not be available due to lack of specialist expertise or resources.
What does the evidence show?
Several clinical trials are taking place across the world, aiming to fully understand the benefits and risks of aHSCT in MS, how it compares to other highly intensive DMTs, and which people with MS might benefit most from this approach.
Clinical trials to date have shown that for relapsing MS, or progressive MS with signs of active inflammation, aHSCT may be able to stop or slow the progression of MS. The therapy is able to reduce new lesions (as seen on an MRI scan) and relapses, and stabilise or improve symptoms. People may see their disability improve, or the worsening of their disability being slowed, but these improvements don’t last for everyone and worsening may occur in future years.
For people with progressive MS who do not show signs of inflammation and who have high levels of disability, current evidence shows that aHSCT is unlikely to be beneficial, and it is associated with increased risk.
In summary, evidence to date suggests the greatest benefits are for people with highly active forms of relapsing MS, who have not responded to, or cannot be treated with, high-efficacy disease modifying therapies. The best candidates are also likely to be under the age of 50, and who are in the earlier, inflammatory stages of MS (had MS for no longer than 10 years, with low to moderate levels of disability).
Crucially, there is a need for further large, gold-standard, randomised controlled trials specifically for MS, which compare aHSCT to currently available MS therapies. The ‘MIST’ trial of aHSCT was run from 2005 to 2016 in the US and other countries and compared aHSCT to standard MS therapies available at the time (which did not include some newer treatments such as alemtuzumab, cladribine and ocrelizumab). Results show that patients receiving AHSCT had improvements in disability scores and significantly fewer relapses compared to those on the standard MS medications. The BEAT-MS study is a Phase III clinical trial in which the safety, efficacy and cost-effectiveness of aHSCT will be directly compared to the best available treatments that have proven benefits for people with active relapsing MS. In Europe there are two Phase III trials taking place comparing aHSCT to alemtuzumab, ocrelizumab, ofatumumab and cladribine (STAR-MS) and to alemtuzumab, ocrelizumab and cladribine (RAM-MS).
Mesenchymal Stem Cell Therapy (MSC therapy)
What is MSC therapy?
Mesenchymal stem cells (MSCs – sometimes referred to as mesenchymal stromal cells or bone marrow stromal cells) are adult stem cells that can produce many different types of cells, including muscle, cartilage and nerve cells. MSC therapy attempts to ‘reformat’ the person’s immune system, and stop the inflammation that contributes to active, relapsing MS. However, these stem cells are not intended to regenerate damaged nerves or other parts of the brain and spinal cord.
MSC therapy does not require chemotherapy, unlike aHSCT. It involves isolating MSCs from the bone marrow. These cells are multiplied in the laboratory (a process that takes a few weeks) and then infused back into the blood or spinal fluid (the fluid surrounding the brain and spinal cord) of the person undergoing therapy. There isn’t enough evidence yet to recommend this type of treatment for people with MS and it should only be offered as part of a clinical trial.
What does the evidence show?
A small number of trials across the world are investigating mesenchymal stem cell therapy in MS. Early research from animal studies and very early stage human trials suggests MSCs could have a positive effect on the immune system. Early phase trials with small numbers of patients did not report any serious adverse events or deaths. However, despite reports and case studies of patients that may have received some early or temporary benefits, there is not yet sufficient evidence to conclude whether there is any benefit or improvement at all in MS. A recent international collaboration, the MESEMS (mesenchymal stem cells for MS) trial, which was partly funded by MSIF, showed that MSC therapy was safe and well tolerated but was not effective at reducing inflammation in people with active forms of multiple sclerosis. For more information click here.
What research is happening into stem cell therapy?
For more detailed information about current research programmes focused on stem cell therapy, including aHSCT, MSC therapy, neural stem cell therapies and other forms of stem cell research, have a look at some of our member organisations’ websites:
- MS Australia
- MS Society of Canada
- Fondation ARSEP (France)
- MS Ireland
- Italian MS Society (AISM)
- MS Society (UK)
- National MS Society (US)
National recommendations/position statements
- Position statement on AHSCT for MS (MS Australia)
- Briefing document and position paper on Stem Cell Treatment (MS Ireland)
- AHSCT in MS (Scientific paper from the National MS Society, US)
- AHSCT in MS (Lay information for people with MS from the National MS Society, US)
- Thérapies par cellules souches dans la SEP (Fondation ARSEP, France)
- Autologe Stammzelltherapie (Schweizerischen Multiple Sklerose Gesellschaft, Switzerland)
- Autologous haematopoietic stem cell transplantation for treatment of multiple sclerosis and neuromyelitis optica spectrum disorder (ECTRIMS and the European Society for Blood and Marrow Transplantation (EBMT) Guidelines)
The information on this page was developed in conjunction with experts from MSIF’s International Medical and Scientific Board and scientific staff from our member organisations. It will be updated regularly as new evidence about stem cell therapies is made available.